
Fillable Online newworldhealthcare Teva Lonquex Referral Form for Amend Proof V2 - newworldhealthcare Fax Email Print - pdfFiller

Immunogenicity assessment of PEGylated proteins, Lonquex, a PEGylated G-CSF case study - ScienceDirect

Teva Pharmaceutical | $TEVA Stock | Shares Rally After Winning FDA Approval for Migraine Therapy - Warrior Trading News
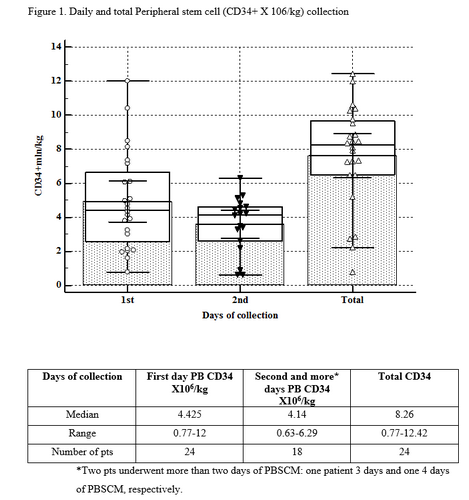
LONG-ACTING G-CSF (LIPEGFILGRASTIM, “LONQUEX”) FOR STEM CELL.... EHA Library. Danylesko I. Jun 15 2019; 267164

Teva Pharmaceutical has been given a green light by the European Commission (EC) for Lonquex, a rival to Amgen's blockbuster Neulasta. | New Drug Approvals








![PDF) Role of lipegfilgrastim in the management of chemotherapy-induced neutropenia [Corrigendum] PDF) Role of lipegfilgrastim in the management of chemotherapy-induced neutropenia [Corrigendum]](https://www.researchgate.net/profile/Louis-Pelus/publication/277615466/figure/fig1/AS:614067458809870@1523416499410/Lipegfilgrastim-is-the-result-of-a-two-step-enzymatic-reaction-Notes-Addition-of-an_Q320.jpg)





